Rapid Assessment of Badea (Passiflora quadrangularis L.) Maturity Degree by Digital Image Analysis and Multivariate Statistical Techniques
DOI:
https://doi.org/10.18779/ingenio.v9i1.1104Keywords:
Maturity index, multivariate analysis, tropical fruit, RGB, PLS-DAAbstract
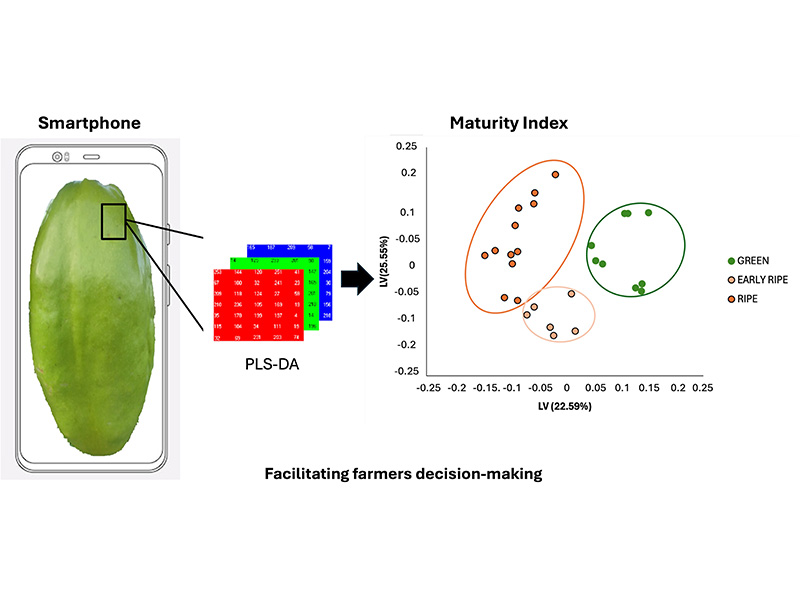
Fruits and vegetables are essential for a healthy diet worldwide, but due to their perishable nature, they are among the most wasted foods. According to the Food and Agriculture Organization (FAO), approximately 45% of food is lost before reaching consumers. Proper handling during and after harvest is crucial to reducing this waste, including identifying the optimal ripening point for both harvest and sale. Passiflora quadrangularis (badea), a traditional South American crop rich in antioxidants, generates significant income for vulnerable communities. Determining the ripeness of badea is key to ensuring better quality and reducing post-harvest losses. In this study, a physicochemical characterization was carried out by days of ripening, and images of the badea were used at three stages of ripeness: green-ripe, early-ripe, and ripe. The physicochemical characterization suggests average pH values of 5,73 and a maturity index of 11,62%, as indicators of an early ripening stage suitable for harvest. These images were analyzed using multivariate statistical methods, including Principal Component Analysis (PCA) and Partial Least Squares Discriminant Analysis (PLS-DA), to identify visual patterns and classify ripeness levels. The model achieved a training accuracy of 100% and a validation accuracy of 83%. These methodologies offer a fast and non-invasive method for assessing fruit ripeness, facilitating informed decisions about the optimal time for harvest and helping to reduce post-harvest waste.
Downloads
References
D. Aune et al., “Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies”, International Journal of Epidemiology, vol. 46, no.3, pp. 1029–1056, Jun. 2017. [Online]. Available: http://doi.org/10.1093/ije/dyw319
Organización de las Naciones Unidas para la Alimentación y la Agricultura (FAO), FAO, El estado mundial de la agricultura y la alimentación 2019: Pérdidas y desperdicio de alimentos en el contexto de sistemas alimentarios sostenibles. 2019. Accessed: Dec. 31, 2024. [Online]. Available: https://openknowledge.fao.org
M. V. Pardo Solórzano, G. M. Costa, and L. Castellanos, “Passiflora By-Products: Chemical Profile and Potential Use as Cosmetic Ingredients,” Sci Pharm, vol. 92, no. 4, pp. 57, Oct. 2024. [Online]. Available: https://doi.org/10.3390/scipharm92040057
A. C. Gazola et al., “The sedative activity of flavonoids from Passiflora quadrangularis is mediated through the GABAergic pathway,” Biomedicine & Pharmacotherapy, vol. 100, pp. 388–393, Apr. 2018. [Online]. Available: https://doi.org/10.1016/j.biopha.2018.02.002
N. Aghilinategh, M. J. Dalvand, and A. Anvar, “Detection of ripeness grades of berries using an electronic nose,” Food Sci Nutr, vol. 8, no. 9, pp. 4919–4928, Sep. 2020. [Online]. Available: https://doi.org/10.1002/fsn3.1788
M. Manzoor, S. B. Hussain, M. A. Anjum, M. Naseer, R. Ahmad, and V. Ziogas, “Effects of Harvest Time on the Fruit Quality of Kinnow and Feutrell’s Early Mandarins (Citrus reticulata Blanco),” Agronomy, vol. 13, no. 3, pp. 802, Mar. 2023. [Online]. Available: https://doi.org/10.3390/agronomy13030802
V. Prasanna, T. N. Prabha, and R. N. Tharanathan, “Fruit Ripening Phenomena–An Overview,” Crit Rev Food Sci Nutr, vol. 47, no. 1, pp. 1–19, Jan. 2007. [Online]. Available: https://doi.org/10.1080/10408390600976841
L. Ortenzi et al., “A Machine Vision Rapid Method to Determine the Ripeness Degree of Olive Lots,” Sensors, vol. 21, no. 9, pp. 2940, Apr. 2021. [Online]. Available: https://doi.org/10.3390/s21092940
V. Paul, R. Pandey, and G. C. Srivastava, “The fading distinctions between classical patterns of ripening in climacteric and non-climacteric fruit and the ubiquity of ethylene—An overview,” J Food Sc. Technol, vol. 49, no. 1, pp. 1–21, Feb. 2012. [Online]. Available: https://doi.org/10.1007/s13197-011-0293-4
A. Payasi, and G. G. Sanwal, “Ripening of climacteric fruits and their control,” J Food Biochem, vol. 34, no. 4, pp. 679–710, Aug. 2010. [Online]. Available: https://doi.org/10.1111/j.1745-4514.2009.00307.x
K. Simkova et al., “Non-destructive and destructive destructive physical measurements as indicators of sugar and organic acid contents in strawberry fruit during ripening,” Sci Hortic, vol. 327, pp. 112843, Mar. 2024. [Online]. Available: https://doi.org/10.1016/j.scienta.2024.112843
M. A. Quelal-Vásconez, M. J. Lerma-García, É. Pérez-Esteve, P. Talens, and J. M. Barat,“Roadmap of cocoa quality and authenticity control in the industry: A review of conventional and alternative methods,” Compr Rev Food Sci Food Saf, vol. 19, no. 2, pp. 448–478, Mar. 2020. [Online]. Available: https://doi.org/10.1111/1541-4337.12522
M. A. Quelal-Vásconez, M. J. Lerma-García, É. Pérez-Esteve, A. Arnau-Bonachera, J. M. Barat, and P. Talens, “Changes in methylxanthines and flavanols during cocoa powder processing and their quantification by near-infrared spectroscopy,” Lwt, vol. 117, pp. 108598, Jan. 2020. [Online]. Available: https://doi.org/10.1016/j.lwt.2019.108598
S. Cubero, N. Aleixos, E. Moltó, J. Gómez-Sanchis, and J. Blasco, “Advances in Machine Vision Applications for Automatic Inspection and Quality Evaluation of Fruits and Vegetables,” Food Bioproc Tech, vol. 4, no. 4, pp. 487–504, May. 2011. [Online]. Available: https://doi.org/10.1007/s11947-010-0411-8
E. Saldaña, R. Siche, M. Luján, and R. Quevedo, “Review: computer vision applied to the inspection and quality control of fruits and vegetables,” Brazilian Journal of Food Technology, vol. 16, no. 4, pp. 254–272, Dec. 2013. [Online]. Available: https://doi.org/10.1590/S1981-67232013005000031
K. Ncama, L. S. Magwaza, A. Mditshwa, and S. Z. Tesfay, “Application of Visible to Near-Infrared Spectroscopy for Non-Destructive Assessment of Quality Parameters of Fruit,” in Infrared Spectroscopy - Principles, Advances, and Applications, IntechOpen, 2019. [Online]. Available: https://doi.org/10.5772/intechopen.80069
S. N. Jha, and S. Gunasekaran, “Authentication of sweetness of mango juice using Fourier transform infrared-attenuated total reflection spectroscopy,” J Food Eng, vol. 101, no. 3, pp. 337–342, Dec. 2010. [Online]. Available: https://doi.org/10.1016/j.jfoodeng.2010.07.019
S. Teerachaichayut, and H. T. Ho, “Non-destructive prediction of total soluble solids, titratable acidity and maturity index of limes by near infrared hyperspectral imaging,” Postharvest Biol Technol, vol. 133, pp. 20–25, Nov. 2017. [Online]. Available: https://doi.org/10.1016/j.postharvbio.2017.07.005
A. A. Bhosale, and K. K. Sundaram, “Nondestructive Method for Ripening Prediction of Papaya,” Procedia Technology, vol. 19, pp. 623–630, 2015. [Online]. Available: https://doi.org/10.1016/j.protcy.2015.02.088
P. P. Subedi, and K. B. Walsh, “Non-invasive techniques for measurement of fresh fruit firmness,” Postharvest Biol Technol, vol. 51, no. 3, pp. 297–304, Mar. 2009. [Online]. Available: https://doi.org/10.1016/j.postharvbio.2008.03.004
Anjali et al., “State-of-the-art non-destructive approaches for maturity index determination in fruits and vegetables: principles, applications, and future directions,” Food Production, Processing and Nutrition, vol. 6, no. 1, pp. 56, Feb. 2024. [Online]. Available: https://doi.org/10.1186/s43014-023-00205-5
G. Vuksani, A. Vuksani, O. Kyçyk, F. Pazari, and T. Thomaj, “Determination of Olive Maturity Stage and Optimal Harvest Interval of ‘Kalinjot’ Cultivar Using Destructive and Non-Destructive Methods,” AgriEngineering, vol. 7, no. 8, pp. 253, Aug. 2025. [Online]. Available: https://doi.org/10.3390/Agriengineering7080253
C. Neupane, M. Pereira, A. Koirala, and K. B. Walsh, “Fruit Sizing in Orchard: A Review from Caliper to Machine Vision with Deep Learning,” Sensors, vol. 23, no. 8, pp. 3868, Apr. 2023. [Online]. Available: https://doi.org/10.3390/s23083868
AOAC, “AOAC Official Method 942.15Acidity (Titratable) of Fruit Products,” in Official Methods of Analysis of AOAC INTERNATIONAL, New York: Oxford University Press, 2023. [Online]. Available: https://doi.org/10.1093/9780197610145.003.3390
D. Kimball, “The Brix/Acid Ratio,” in Citrus Processing, Dordrecht: Springer Netherlands, 1991, pp. 55–65. [Online]. Available: https://doi.org/10.1007/978-94-011-3700-3_4
M. E. Arena, I. S. Povilonis, V. Borroni, D. Constenla, and S. Radice, “Changes in physicochemical properties at different development stages of Hexachlamys edulis fruit, an underutilized South American species,” Heliyon, vol. 7, no. 11, pp. e08323. Nov. 2021. [Online]. Available: https://doi.org/10.1016/j.heliyon.2021.e08323
S. D. Ramaiya, H. A. Ismail, N. Khairuddin, and M. A. Awang, “Ripening Characterization, Fruit Quality and Sensory Attributes of Passiflora quarangularis L.,” International Journal of Chemical and Biochemical Sciences, vol.24, no.6, pp. 925-936, 2023. [Online]. Available: https://www.iscientific.org/wp-content/uploads/2024/01/106-ijcbs-23-24-6-106.pdf
J. M. Sánchez Soto, L. H. Ramos, and E. J. Torres Mendoza, “Efectos fisiológicos de badea (Passiflora quadrangularis) y yuca (Manihot esculenta) utilizando recubrimientos a base de cera y parafina bajo conservación en frío.,” Revista colombiana de investigaciones agroindustriales, vol. 1, no. 1, pp. 33-43, Dec. 2014. [Online]. Available: https://doi.org/10.23850/24220582.113
S. D. Ramaiya, H. H. Lee, Y. J. Xiao, N. S. Shahbani, M. H. Zakaria, and J. S. Bujang, “Organic cultivation practices enhanced antioxidant activities and secondary metabolites in giant granadilla (Passiflora quadrangularis L.),” PLoS One, vol. 16, no. 7, Jul. 2021. [Online]. Available: https://doi.org/10.1371/journal.pone.0255059
Simkova, K., Veberic, R., Hudina, M., Grohar, M. C., Pelacci, M., Smrke, T., Ivancic, T., Cvelbar Weber, N., and Jakopic, J. (2024b). Non-destructive and destructive physical measurements as indicators of sugar and organic acid contents in strawberry fruit during ripening. Scientia Horticulturae, vol. 327, Mar. 2024. [Online]. Available: https://doi.org/10.1016/j.scienta.2024.112843
D. Escobar Figueroa, and E. Roa Guerrero, “Sistema de visión artificial para la identificación del estado de madurez de frutas (granadilla),” Redes de Ingeniería, vol. 7, no. 1, pp. 78, Jun. 2016. [Online]. Available: https://doi.org/10.14483/udistrital.jour.redes.2016.1.a08
S. Tu, Y. Xue, C. Zheng, Y. Qi, H. Wan, and L. Mao, “Detection of passion fruits and maturity classification using Red-Green-Blue Depth images,” Biosyst Eng, vol. 175, pp. 156–167, Nov. 2018. [Online]. Available: https://doi.org/10.1016/J.Biosystemseng.2018.09.004
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Luis Egas, Gina Guapi, Maribel Quelal-Vásconez

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Licensing Agreement
This journal provides free access to its content through its website following the principle that making research available free of charge to the public supports a larger exchange of global knowledge.
Web content of the journal is distributed under a Attribution-NonCommercial-ShareAlike 4.0 International.
Authors may adopt other non-exclusive license agreements for the distribution of the version of the published work, provided that the initial publication in this journal is indicated. Authors are allowed and recommended to disseminate their work through the internet before and during the submission process, which can produce interesting exchanges and increase citations of the published work.





